The U.S. Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) issued a Regulatory Status Review (RSR) on September 7, 2022, stating that the new genetically modified (GM) purple tomato from Norfolk Plant Sciences may be safely grown and used in breeding in the United States.
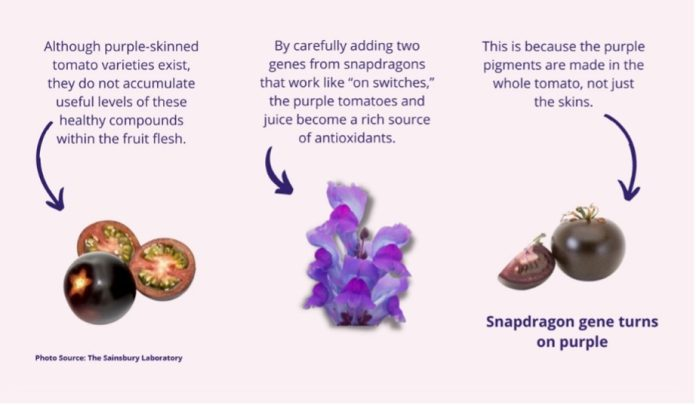
The purple tomato was modified to alter its color and enhance its nutritional quality. Domesticated tomatoes already have genes to produce anthocyanins, but they are not “turned on” in most fruits. The GM purple tomatoes have two genes added from snapdragons that work like “on switches,” so the fruits and juice are a rich source of antioxidants because purple pigments are made in the whole tomato, not just the skins. In the review, USDA APHIS has determined that the GM tomato is unlikely to pose an increased plant pest risk relative to other cultivated tomatoes and is not subject to regulations limiting “Movement of Organisms Modified or Produced through Genetic Engineering” (7 CFR part 340).
The USDA APHIS RSR is based on information from Norfolk Plant Sciences and their familiarity with tomato varieties, knowledge of the traits that alter fruit color and nutritional quality, and understanding of the modifications. USDA APHIS issued a response letter indicating the plant is not subject to regulation. This is the first RSR response that USDA APHIS has issued under the revised biotechnology regulations.
Professor Cathie Martin, who developed the anthocyanin-rich purple tomato in 2008 said, “This is fantastic, I never thought I would see this day. We are now one step closer to my dream of sharing healthy purple tomatoes with the many people excited to eat them.”- Crop Biotech Update
Article originally published in: https://thepetridish.my/2023/02/14/gm-purple-tomato-gets-approval-in-the-united-states/